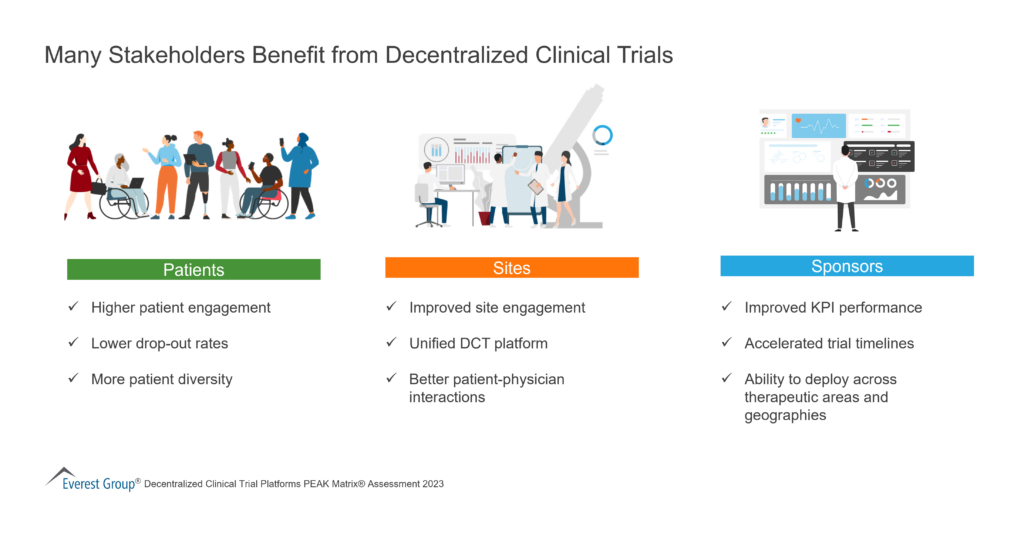
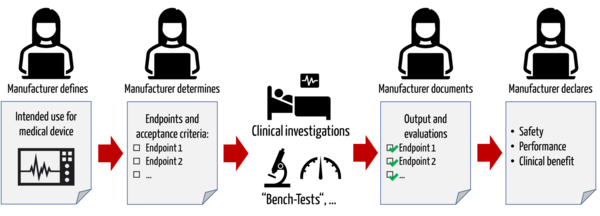
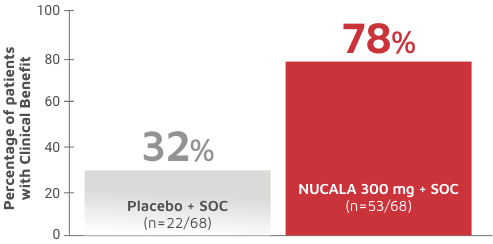
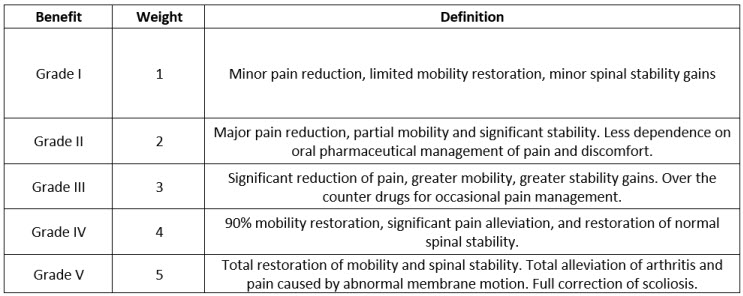
CodeBreak 100: results show durability of clinical benefit with sotorasib in patients with non-small cell lung cancer - Onco Americas

Cancers | Free Full-Text | Mutations Associated with No Durable Clinical Benefit to Immune Checkpoint Blockade in Non-S-Cell Lung Cancer

Choice of Primary (or Co-primary) Endpoints: Efficacy, Safety, or Net Clinical Benefit in Superiority and Non-inferiority Trials | tctmd.com

Net clinical benefit of anticoagulation for atrial fibrillation following intracerebral hemorrhage - Matthew A Pappas, James F Burke, 2020
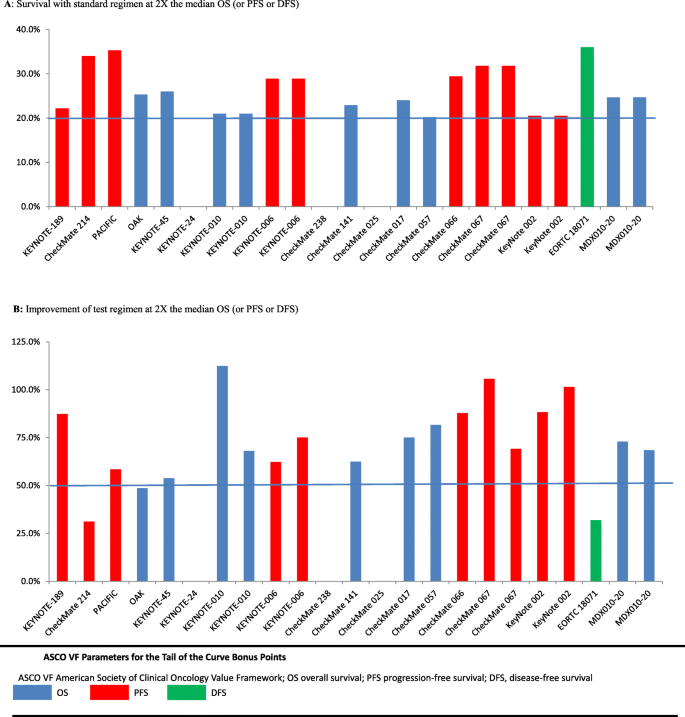
Clinical benefit of immune checkpoint inhibitors approved by US Food and Drug Administration | BMC Cancer | Full Text

Patient Global Impression of Benefit–Risk (PGI-BR): Incorporating Patients' Views of Clinical Benefit–Risk into Assessment of New Medicines | SpringerLink

Pathological complete response and long-term clinical benefit in breast cancer: the CTNeoBC pooled analysis - The Lancet

FDA allows drugs without proven clinical benefit to languish for years on accelerated pathway | The BMJ

Antagonizing CD105 and androgen receptor to target stromal-epithelial interactions for clinical benefit: Molecular Therapy