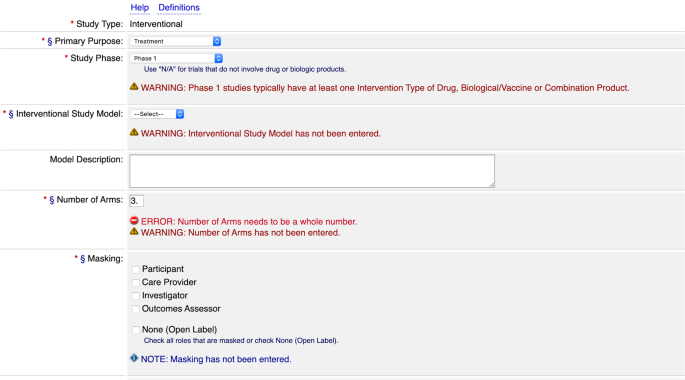
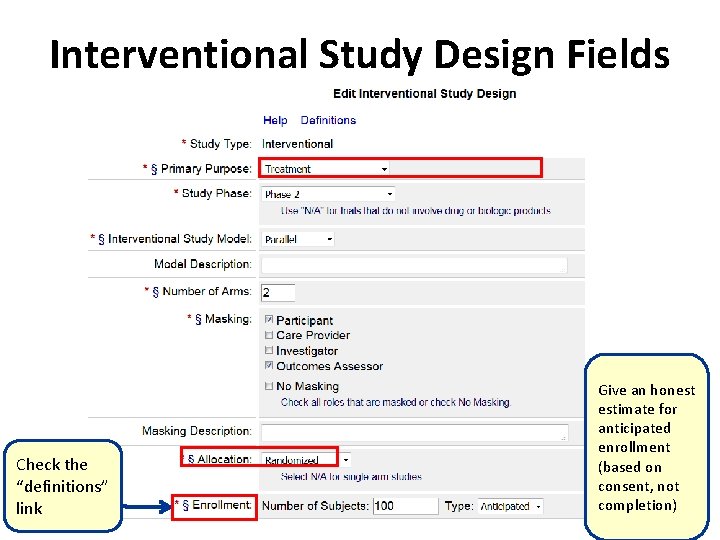
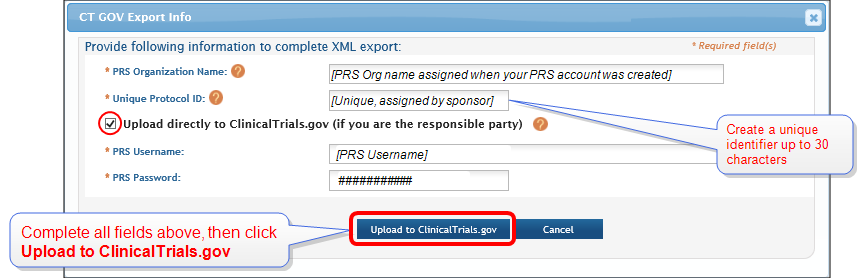
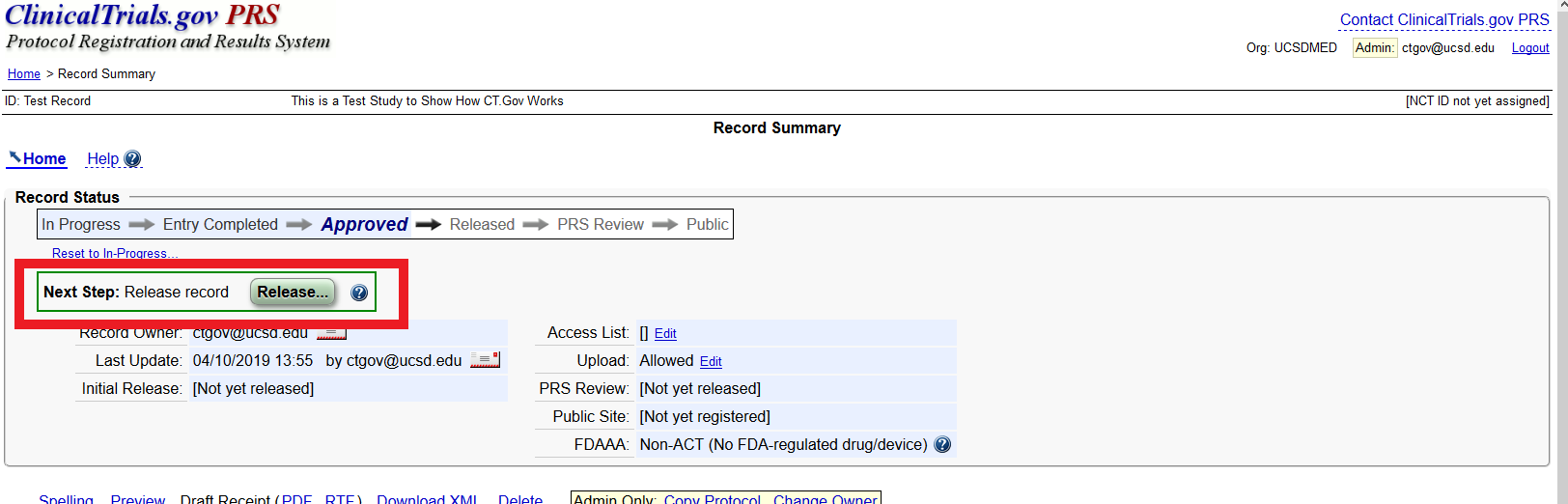
PDF) ClinicalTrials.gov Protocol Registration and Results System (PRS) Receipt Study Identification Unique Protocol ID: AlAzharPGB Brief Title: Topical Preparation of Pomegranate and Ginkgo Biloba for Anti-aging Activities Official Title: Formulation ...

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

ClinicalTrials.gov trial registration submission cycle. JHUSOM: Johns... | Download Scientific Diagram

PDF) 3381 Reducing Problem Records in the Johns Hopkins University ClinicalTrials.gov Protocol Registration and Results System (PRS)